 |
| Home | ON2.2. Group | ON2. Division | Institute Home |
|
The influence of mean-free path in vapour, temperature and composition of liquid upon evaporation at nano and microscalePrincipal Investigator: Daniel Jakubczyk, Ph.D., D.Sc., Eng.Research project objectivesMass
and energy transport phenomena at the liquid–gas interface (e.g.
evaporation) has been be-yond correct analytical description for more
than 100 years (journal paper published together by IPC PAS and IP PAS
[Rep. Prog. Phys., 76, 034601 (2013)] IF=14.937). Our collaborators
from IPC PAS discovered that under low pressure, transport of heat to
evaporating droplets is limited and additionally, a temperature drop
appears on the phase boundary [Phys. Rev. Lett, 100, 055701 (2008)]. In
IP PAS we have built unique scientific instruments to measure
evaporation of freely suspended droplets of micrometric sizes [J. Phys.
Chem. A, 112, 5152 (2008)]. Collaboration be-tween our research groups
has already resulted in important publications, e.g. [Rep. Prog. Phys.,
76, 034601 (2013)]. In our work [Soft Matter, 9, 7766 (2013)], we
merged molecular dynamic (MD) computer simulations of the research
group from IPC PAS with experimental results obtained from levitating
microdroplets of water, glycols and glycerine performed at IP PAS, and
we proposed an analytical equation describing evaporation of droplets.
We tested it for 11 orders of magnitude in the time scale and 4 orders
of magnitude in radius. Our description requires only two parame-ters:
the first is related to temperature drop during evaporation, while the
second is responsible for description of particles mean free path in
vapour surrounding droplet. Given equation has been veri-fied
experimentally for standard (room) temperature at atmospheric pressure.
On the other hand, our partners from IPC PAS in their work [J. Chem.
Phys., 130, 074707 (2009)] also investigated nu-merically the rules
governing evaporation of liquids in vacuum. The goal of the current
project is to describe quantitatively evaporation in the middle range
of pressures, i.e. between atmospheric pres-sure and vacuum, and in
large range of temperatures. We plan to change particles mean free path
in vapour at least by 3 orders of magnitude (from ~70 nm at normal
conditions up to ~100 μm) by decreasing the ambient pressure. We will
also test the proposed description as a function of tem-perature from
~230 K to ~330 K (also in the vicinity of the triple point) and
chemical composition of droplets. Necessary computer simulations will
be carried out at IPC PAS as the complement to physical experiments of
IP PAS. Research methodology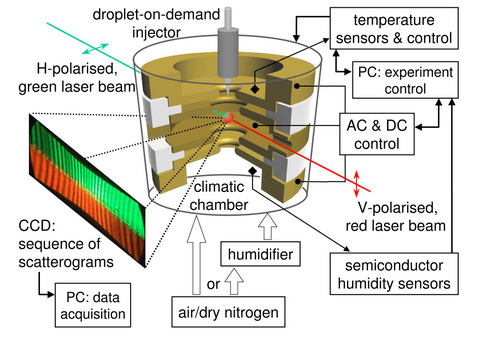 In the physical experiment we will measure evolution of freely
suspended, evaporating droplets with various volatility, molecular
weight (glycerine, glycols, water) and composition under different
temperature conditions, pressure and composition of ambient atmosphere.
We will use the experimental setup presented in Figure beside. Droplet
evolution will be precisely measured with methods based on the light
scattering and electrostatic weighting developed at IP PAS. Scientific
equipment built at IP PAS (see Figure) will be modified to conduct
measurements under lower pressure and in vacuum. Additionally, surfaces
of liquid will be analysed with dielectric and metallic inclusions. In
that case, measurements will be performed for droplets of suspensions. In the physical experiment we will measure evolution of freely
suspended, evaporating droplets with various volatility, molecular
weight (glycerine, glycols, water) and composition under different
temperature conditions, pressure and composition of ambient atmosphere.
We will use the experimental setup presented in Figure beside. Droplet
evolution will be precisely measured with methods based on the light
scattering and electrostatic weighting developed at IP PAS. Scientific
equipment built at IP PAS (see Figure) will be modified to conduct
measurements under lower pressure and in vacuum. Additionally, surfaces
of liquid will be analysed with dielectric and metallic inclusions. In
that case, measurements will be performed for droplets of suspensions.Research Project impactNumerous
physical phenomena at macroscale are governed by evaporation of liquids
in gases at micro and nanoscales. For instance, evaporation of oceans
is mainly caused by evaporation of droplets atomised by wind from water
surface. 80-90% of water vapour (main contributor of the greenhouse
effect) in atmosphere comes from the oceans. Therefore, understanding
of evaporation processes is needed to develop multiscale models of
weather and climate. Currently, several re-search groups are working on
cooling of electronic microcircuits by spraying out droplets of
liquids. Similarly, fuels are transported to combustion chamber of
engines as microdroplets. Their evapora-tion absorbs significant part
of heat released by fuel burning and ultimately this process is
govern-ing engines’ efficiency. Bibliography[1]. Rep. Prog. Phys., vol. 76 (3), pp. 034601, 2013.[2]. Phys. Rev. Lett, 100, 055701 (2008). [3]. J. Phys. Chem. A, 112, 5152 (2008). [4]. Soft Matter, vol. 9, pp. 7766, 2013. [5]. J. Chem. Phys., 130, 074707 (2009). Publications
|
| All rights reserved. |